Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yamashita, Shinichi*; Iwamatsu, Kazuhiro; Maehashi, Yuki*; Taguchi, Mitsumasa; Hata, Kuniki; Muroya, Yusa*; Katsumura, Yosuke*
RSC Advances (Internet), 5(33), p.25877 - 25886, 2015/02
Times Cited Count:12 Percentile:37.81(Chemistry, Multidisciplinary)Pulse radiolysis experiments were carried out to observe transient absorptions of reaction intermediates produced in N O
O and Ar-saturated aqueous solutions containing 0.9-900 mM NaBr. The most important species among the reaction intermediates are BrOH
and Ar-saturated aqueous solutions containing 0.9-900 mM NaBr. The most important species among the reaction intermediates are BrOH 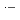 and Br
and Br
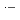 , which commonly have absorption peaks around 360 nm. The experimental results were compared with the results of simulation based on a spur diffusion model. Each of several complicated sequential radiation-induced chemical reactions was carefully considered, optimizing its rate constant within a range of reported values. All the experimental results were able to be universally reproduced by the simulation, assuming a reaction not yet reported, 2BrOH
, which commonly have absorption peaks around 360 nm. The experimental results were compared with the results of simulation based on a spur diffusion model. Each of several complicated sequential radiation-induced chemical reactions was carefully considered, optimizing its rate constant within a range of reported values. All the experimental results were able to be universally reproduced by the simulation, assuming a reaction not yet reported, 2BrOH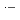
 Br
Br + 2OH
+ 2OH , with a rate constant of 3.8
, with a rate constant of 3.8  10
10 M
M s
s , which is significant only within 10 micro-s for rather high bromide concentrations (
, which is significant only within 10 micro-s for rather high bromide concentrations ( 10 mM). Primary
10 mM). Primary 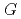 values, which are yields after sufficient diffusion from the spur to the perimeter region during 100 ns, of major water decomposition products, as well as of the reaction intermediates, were calculated for N
values, which are yields after sufficient diffusion from the spur to the perimeter region during 100 ns, of major water decomposition products, as well as of the reaction intermediates, were calculated for N O
O and Ar-saturated conditions as a function of NaBr concentration.
and Ar-saturated conditions as a function of NaBr concentration.
Taguchi, Mitsumasa
Hoshasen Kagaku, (77), p.2 - 7, 2004/05
no abstracts in English
Working Group in the Advisory Committee of the Analytical Studies for Application of Charged Particl
JAERI-Review 2003-008, 42 Pages, 2003/03
The position in the research field of radiation application of "Theoretical Analysis Group for Radiation Application" which will be set up within fiscal 2003, and the relation between the research that this analytical group will advance in future and the analytical research made so far at JAERI Takasaki are summarized. First, the theoretical analysis group for radiation application extracts only the core element from the analysis of reactive process of charged particles in the materials and from the irradiation effect, and then constructs the simplified model. Second, the validity of the constructed model is verified by advancing the theoretical analysis based on the model, and the calculated result is fed back to the experiment, and then the verification is carried out. Finally, the general-purpose theorization will be conducted by generalizing and universalizing the obtained model concerning the specific system. The obtained outcomes will be applied to other systems, and, will consequently, contribute to the promotion of new technological development.
Tachimori, Shoichi; *
JAERI-Data/Code 96-030, 116 Pages, 1996/10
no abstracts in English
 /N
/N O mixture in the presence of molecular sieve 5A
O mixture in the presence of molecular sieve 5AShimizu, Yuichi; Nagai, Shiro
Applied Radiation and Isotopes, 41(5), p.457 - 461, 1990/00
no abstracts in English
Shimizu, Yuichi; Nagai, Shiro
Radiation Physics and Chemistry, 36(6), p.763 - 766, 1990/00
no abstracts in English
Shimizu, Yuichi
Radiation Physics and Chemistry, 36(3), p.291 - 294, 1990/00
no abstracts in English
Hoken Butsuri, 22, p.189 - 207, 1987/00
no abstracts in English
Hayakawa, Naohiro; Kuriyama, Isamu
Journal of Polymer Science; Polymer Chemistry Edition, 14(6), p.1513 - 1517, 1976/06
no abstracts in English
Kumada, Takayuki
no journal, ,
no abstracts in English
Hirade, Tetsuya
no journal, ,
We have measured water by the positron annihilation age-momentum correlation (AMOC). As a result, a spin conversion reaction between triplet positronium (o-Ps; positronium is a bonded state of an electron and a positron) and an OH radical was observed. From its temperature dependence, the reaction of the spin-correlated pairs by the "cage effect" was significant at temperatures below 10 C. In addition, it has been shown that there are cases where this "cage effect" does not disappear even at 10
C. In addition, it has been shown that there are cases where this "cage effect" does not disappear even at 10 C or higher when the temperature is raised. This indicates the possibility that a structure that causes a "cage effect" is stabilized at temperatures below 10
C or higher when the temperature is raised. This indicates the possibility that a structure that causes a "cage effect" is stabilized at temperatures below 10 C. The surface of the sample is in contact with the air. The pH measurement indicated that carbon dioxide dissolved in the sample was related to this structure. This indicates the possibility that carbon dioxide is kept in water up to about 25
C. The surface of the sample is in contact with the air. The pH measurement indicated that carbon dioxide dissolved in the sample was related to this structure. This indicates the possibility that carbon dioxide is kept in water up to about 25 C.
C.